Menu
Close
Clinical studies carried out by the UNIVERSITY of PAVIA through Clinical and Instrumental Evaluation of the two formulations, YALODÉ COMPRESSES, and YALODÉ INTENSIVE WRINKLE REPAIR have unequivocally demonstrated the Efficacy and Tolerability of the YALODÉ Line products.
Specifically, the 2 clinics performed by dermatologists on a sample of 20 women showed that the anti-aging tablets and face cream, in addition to safety tests on all products in the Line, were well tolerated in 100% of subjects.

CLINICAL/INSTRUMENTAL EVALUATION OF THE MOISTURIZING AND ANTI-WRINKLE EFFICACY OF A DIETARY SUPPLEMENT
SKIN PROFILOMETRY ASSESSMENT
Skin relief was assessed by PRIMOS 3D. It is a 3D scanner that acquires a point cloud in a three-dimensional coordinate system of the object’s surface.
| T30 | T60 | |
|---|---|---|
| Average % change vs T0 | -9,1% | -12,8% |
| T test vs T0 | 0,010 | 0,000 |
| Max (maximum variation) | -34,1% | -35,3% |
| Min (minimum change) | 22,4% | 11,2% |
| % of subjects in whom instrumental reduction in wrinkle depth was recorded (< of -1%) | 65%* | 85%** |
TABLE 1. The table shows the average, minimum and maximum percentage changes in wrinkle depth analyzed after 30 and 60 days of daily product use.
* 13 subjects – ** 17 subjects
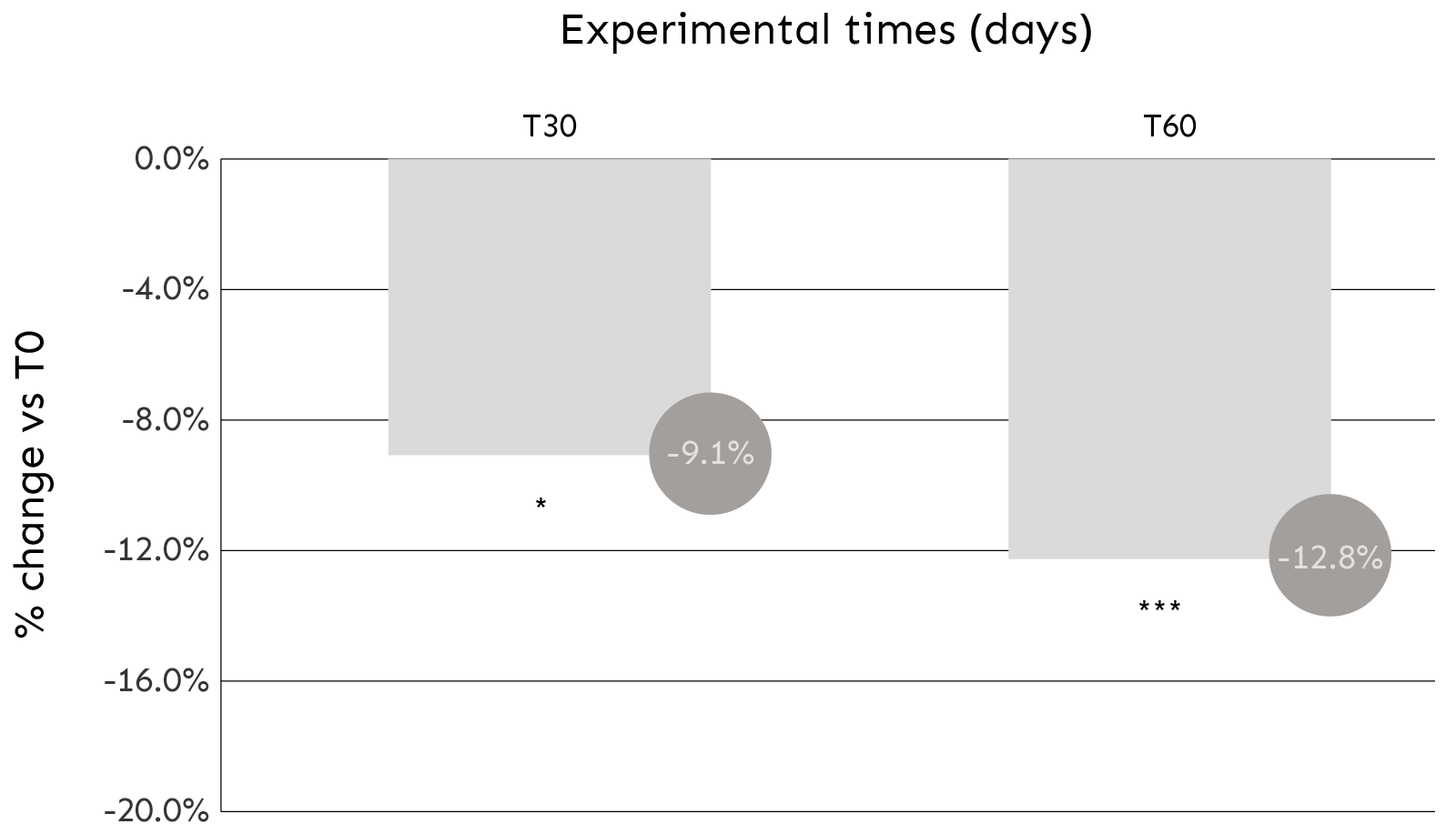
COMMENT: After 30 and 60 days of product use, there was a significant improvement in skin roughness (expressed as an average % reduction in wrinkle depth analyzed) of 9.1% and 12.8%, respectively.
NOTE: Intra-group statistical analysis (vs. T0) is shown in the graph.
LEGEND:
*** ,p<0.001
** ,p<0.01
* ,p<0.05
ASSESSMENT OF SKIN HYDRATION
Hydration measurement is based on the internationally recognized CORNEOMETER® method (Courage+Khazaka electronic GmbH).
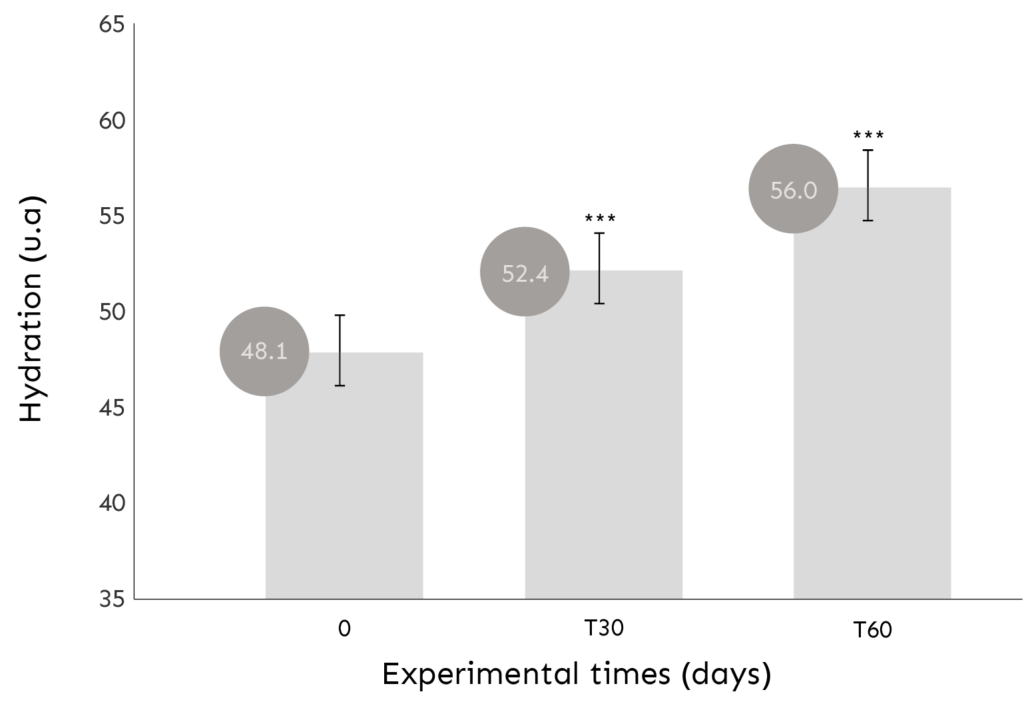
COMMENT: After 30 and 60 days of product use, there was a significant improvement in skin hydration of 9.6 percent and 17.4 percent, respectively (mean values).
NOTE: Intra-group statistical analysis (vs. T0) is shown in the graph.
LEGEND:
*** ,p<0.001
** ,p<0.01
* ,p<0.05
Conclusions:

CLINICAL/INSTRUMENTAL EVALUATION OF THE MOISTURIZING AND
ANTI-WRINKLE EFFICACY OF A COSMETIC PRODUCT
CUTAN ELASTICITY ASSESSMENT
Evaluation of the R2 PARAMETER through the MPA 580 CUTOMETER.
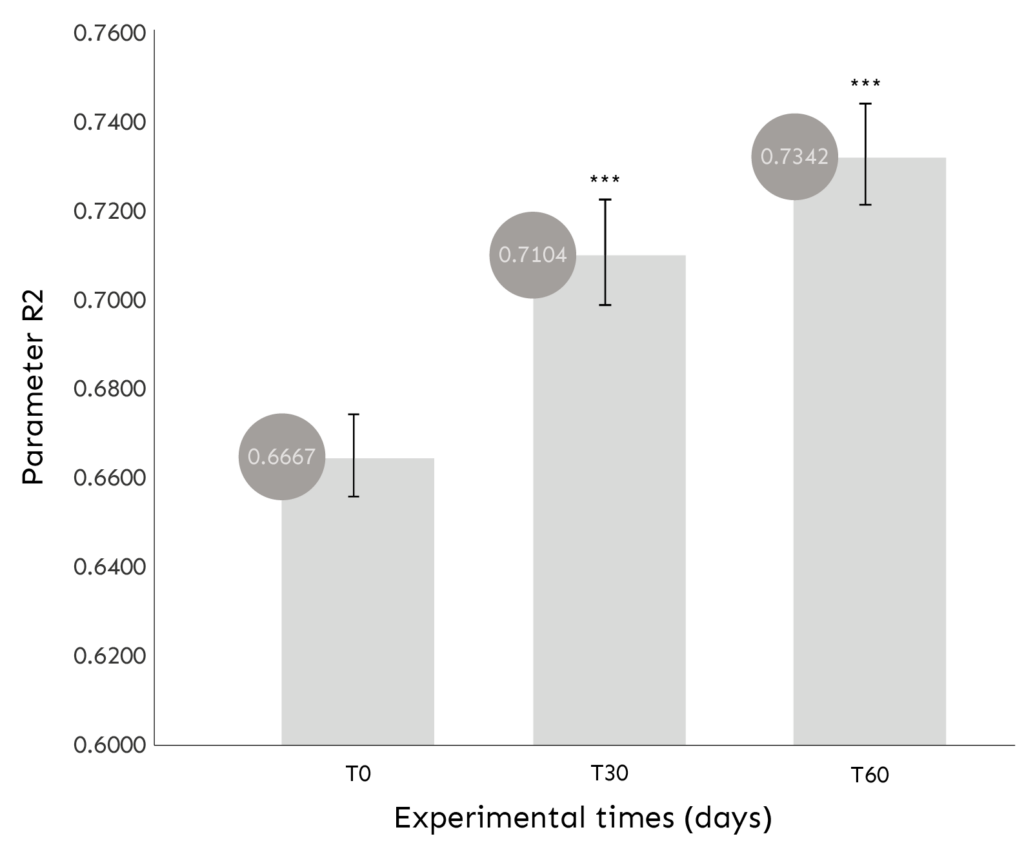
COMMENT: The product results, at all monitored experimental times, in a statistically significant (p<0.001) increase in skin elasticity.
NOTE: Above the error bar is the intra-group statistical analysis (vs. T0).
LEGEND:
*** ,p<0.001
SKIN PROFILOMETRY ASSESSMENT
| T30 | T60 | |
|---|---|---|
| Average % change vs T0 | -9,0% | -15,3% |
| T test vs T0 | 0,001 | 0,000 |
| Max (maximum variation) | -28,8% | -37,5% |
| Min (minimum change) | 2,1% | 3,1% |
| % of subjects in whom instrumental reduction in wrinkle depth was recorded (< of -1%) | 75%* | 85%** |
* 15 subjects – ** 17 subjects

COMMENT: The product results, at all monitored experimental times, in a statistically significant (p<0.001) reduction in wrinkle depth analyzed.
NOTE: Intra-group statistical analysis (vs. T0) is shown in the graph.
ASSESSMENT OF SKIN HYDRATION INDEX

COMMENT: The product results, at all monitored experimental times, in a statistically significant (p<0.001) increase in skin hydration.
NOTE: Above the error bar is the intra-group statistical analysis (vs.T0).
LEGEND:
*** ,p<0.001
Conclusions:
Increased skin elasticity (parameter R2) after both 30 days (+7.0%) and 60 days (+10.7%) of use;
Improvement in the profilometric parameter analyzed (reduction of wrinkle depth in the periocular area) after both 30 days (-9.0%) and 60 days (-15.3%) of use;
Increased skin hydration after both 30 days (+21.2%) and 60 days (+25.8%) of use.

CLINICAL-INSTRUMENTAL ASSESSMENT OF THE EFFICACY OF A COSMETIC PRODUCT COADJUVANT THE TREATMENT OF THE CELLULITE-DERIVED SKIN IMPERFECTIONS
EVALUATION OF SKIN THERMOGRAPHY
The graph shows percentage of subjects related to the effect.
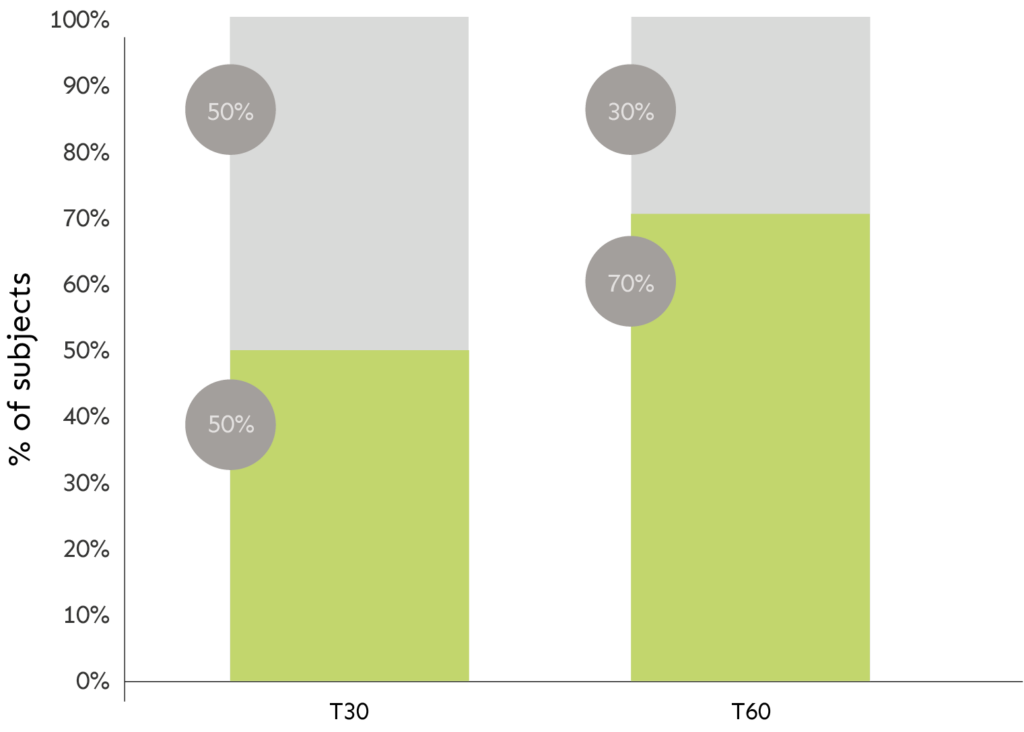
LEGEND:
ASSESSMENT OF THE THICKNESS OF LOCALIZED FAIRY TISSUE
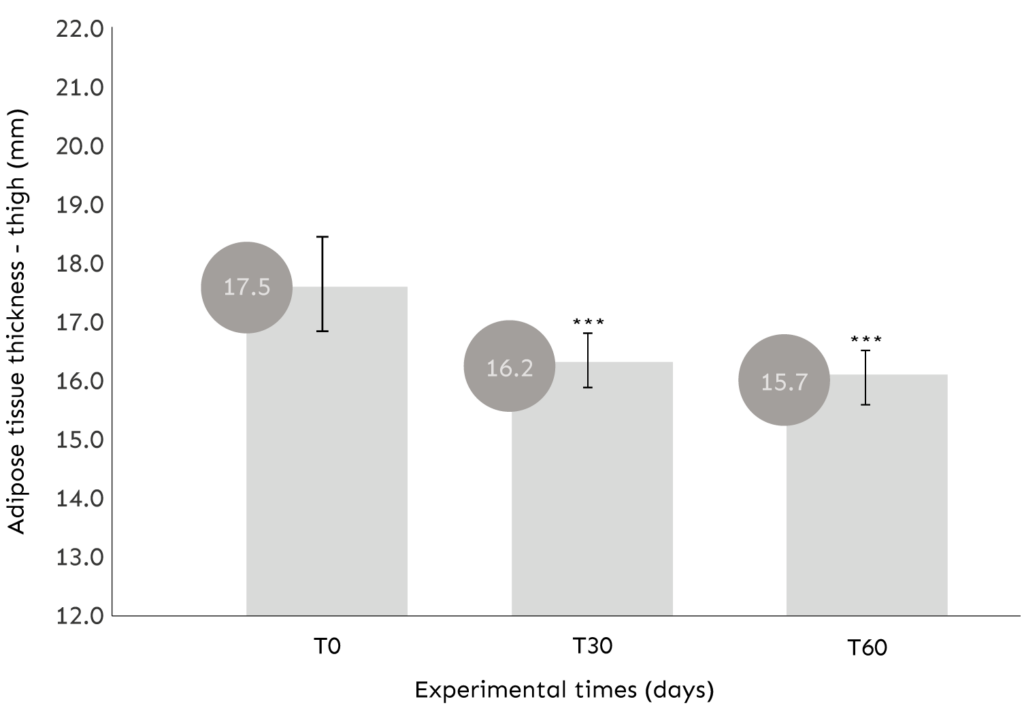
COMMENT: After 30 and 60 days of use, the product determines a statistically significant (p<0.001) reduction of the adipose tissue thickness of the thigh.
LEGEND:
*** ,p<0.001
ASSESSMENT OF SKIN ELASTICITY
Evaluation of the R2 PARAMETER through the MPA 580 CUTOMETER.
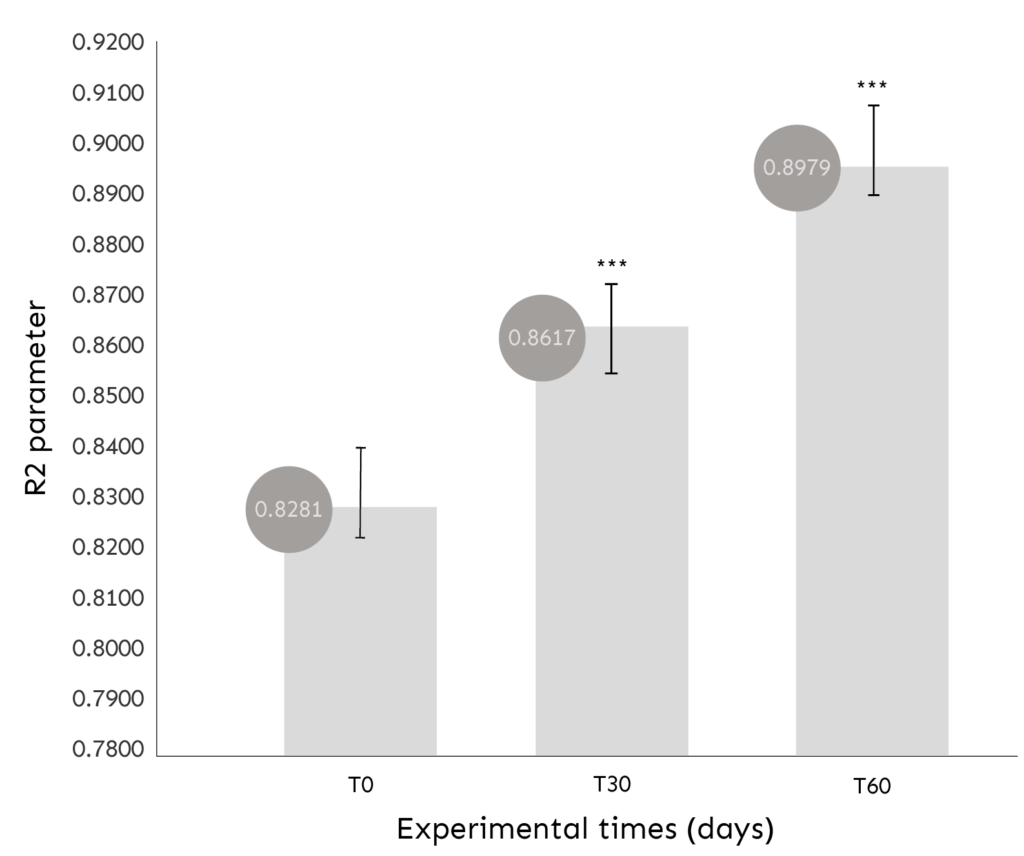
COMMENT: After 30 and 60 days of use, the product determines a statistically significant (p<0.001) improvement of skin elasticity.
LEGEND:
*** ,p<0.001
ASSESSMENT OF THIGH CIRCUMFERENCE
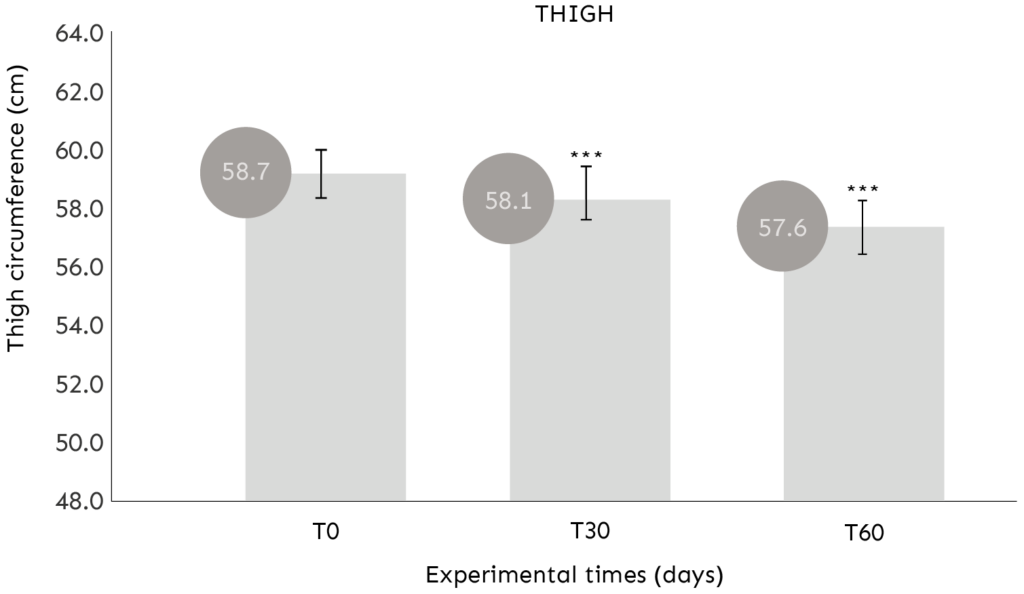
COMMENT: After 30 and 60 days of use, the product determines a statistically significant (p<0.001) reduction of the thigh circumference.
LEGEND:
*** ,p<0.001
ASSESSMENT OF GLUTEUS CIRCUMFERENCE
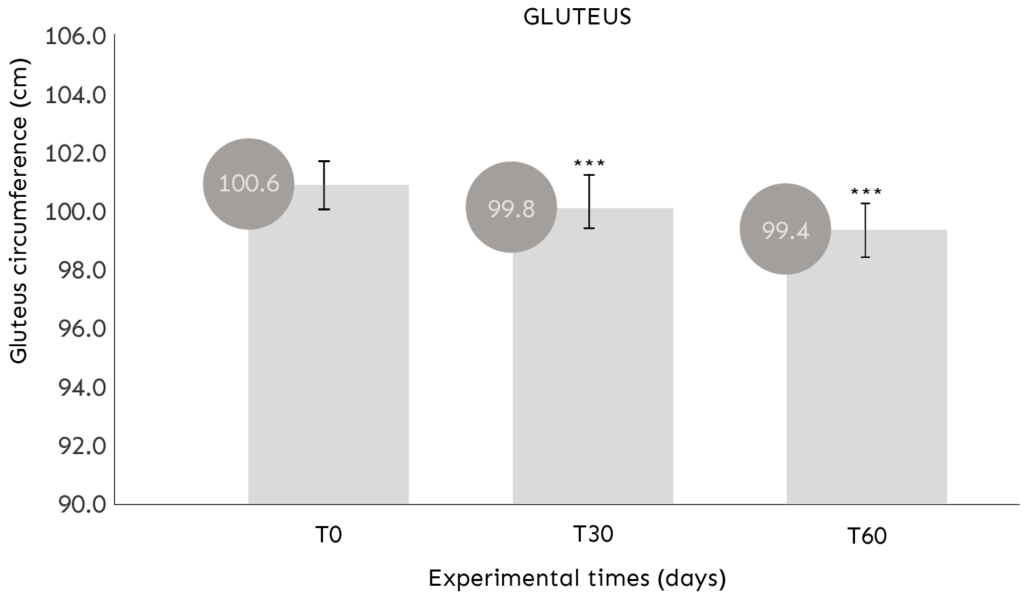
COMMENT: After 30 and 60 days of use, the product determines a statistically significant (p<0.001) reduction of the gluteus circumference.
LEGEND:
*** ,p<0.001
CLINICAL ASSESSMENT OF SKIN SMOOTHNESS
The graph shows percentage of subjects related to the effect.
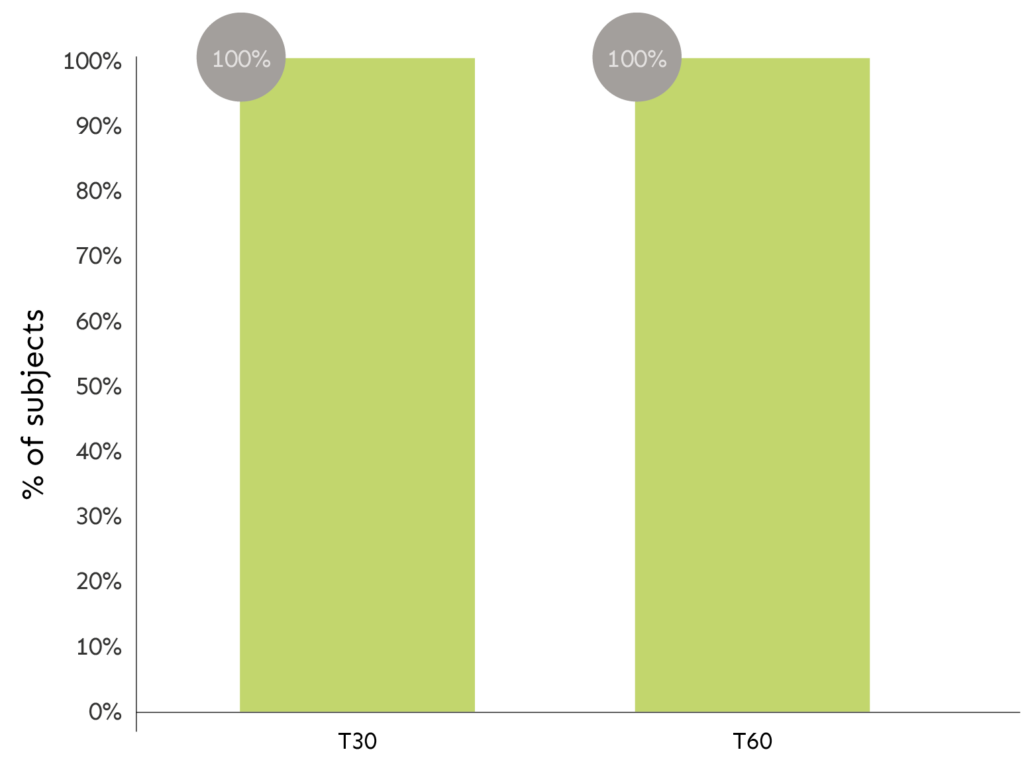
LEGEND:
CLINICAL EVALUATION OF THE “ORANGE PEEL” SKIN APPEARANCE
The graph shows percentage of subjects related to the effect.
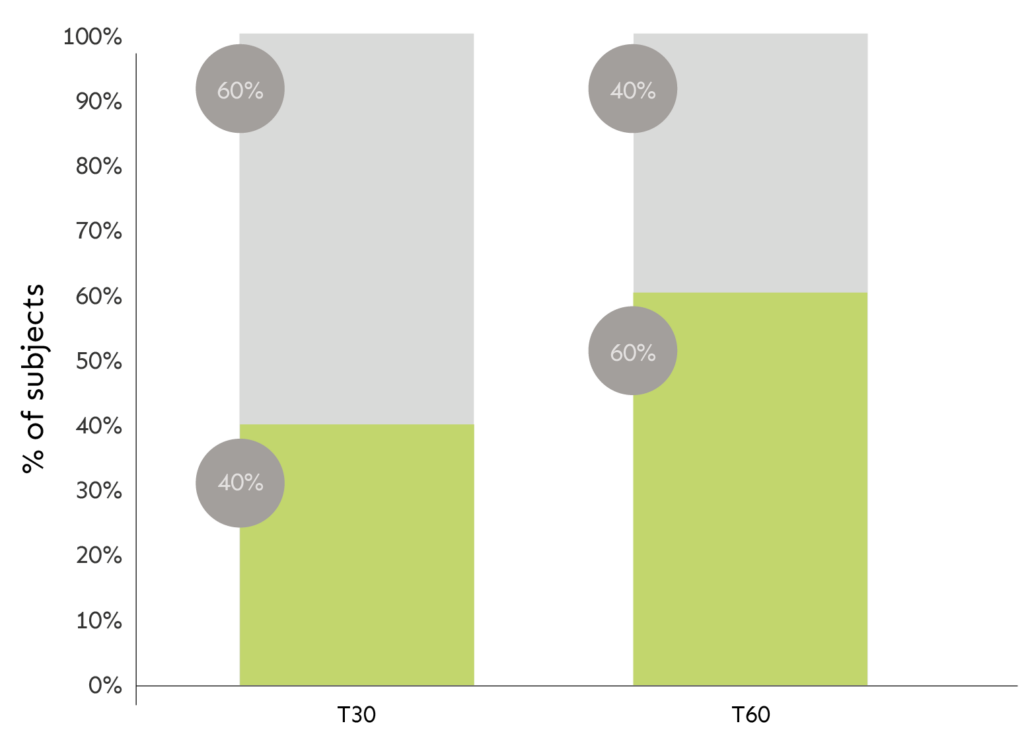
LEGEND:
Conclusions:
After 60 days of use Syaloderm Polyenzymatic Gel determines an improvement in all monitored instrumental parameters.
Particularly, the following were recorded:
Average reduction in the thickness of the subcutaneous fat of the thigh of -1.31mm (T30) and -1.87mm (T60);
Improvement in skin elasticity of +4.1% (T30) and +8.5% (T60);
Average reduction in thigh circumference of -0.61cm (T30) and -1.03cm (T60);
Average reduction in gluteus circumference of -0.85cm (T30) and -1.24cm (T60).
Monday to Friday
08:30–12:30 / 13:30–17:30 (Italian time zone)
Mail: info@sialormilano.it
© 2024, Sialor Milan – Designed by Red Velvet Studio Srl